Climedo for clinics and research institutes
Climedo for Hospitals and Research Institutes
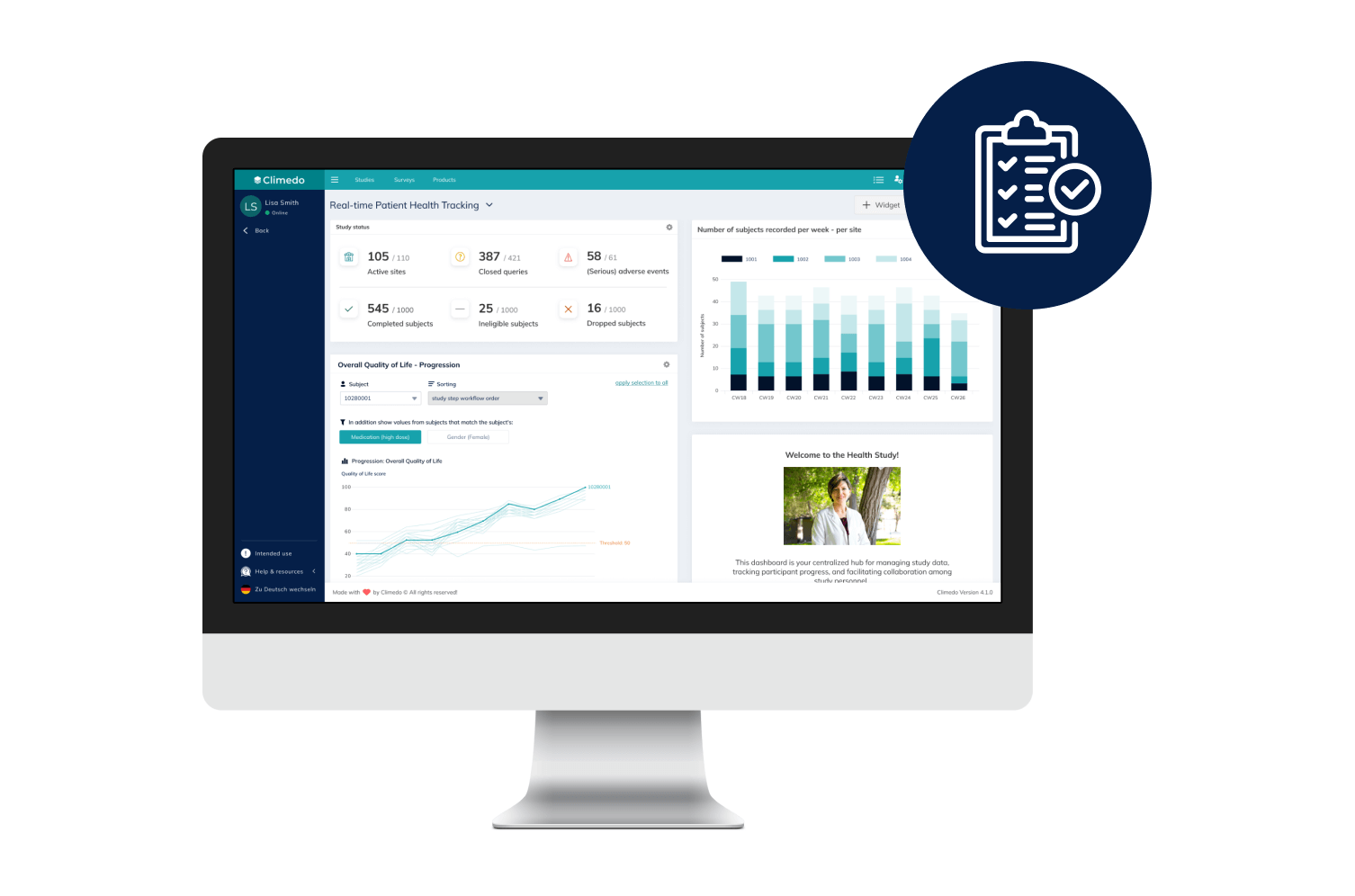
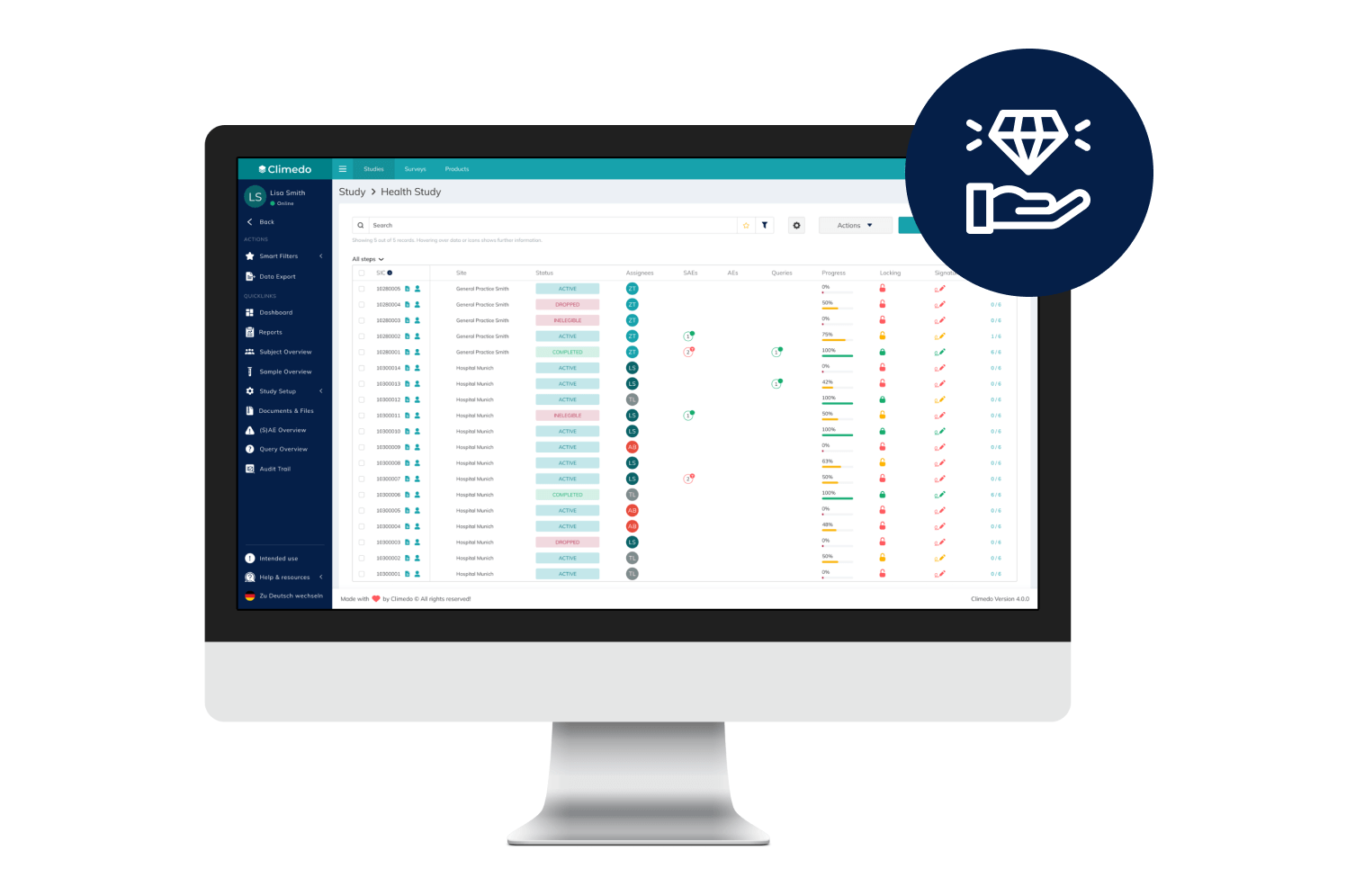
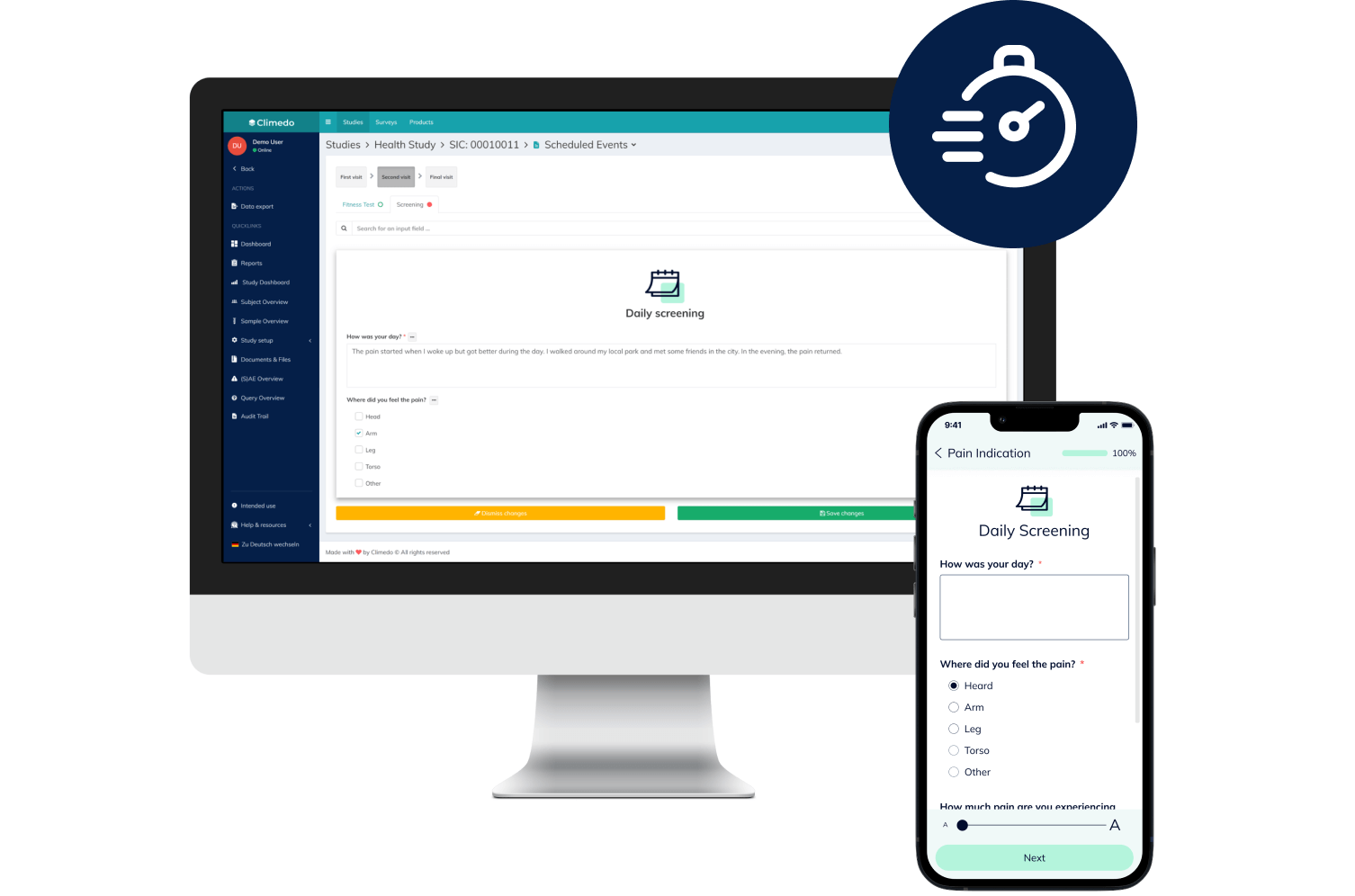
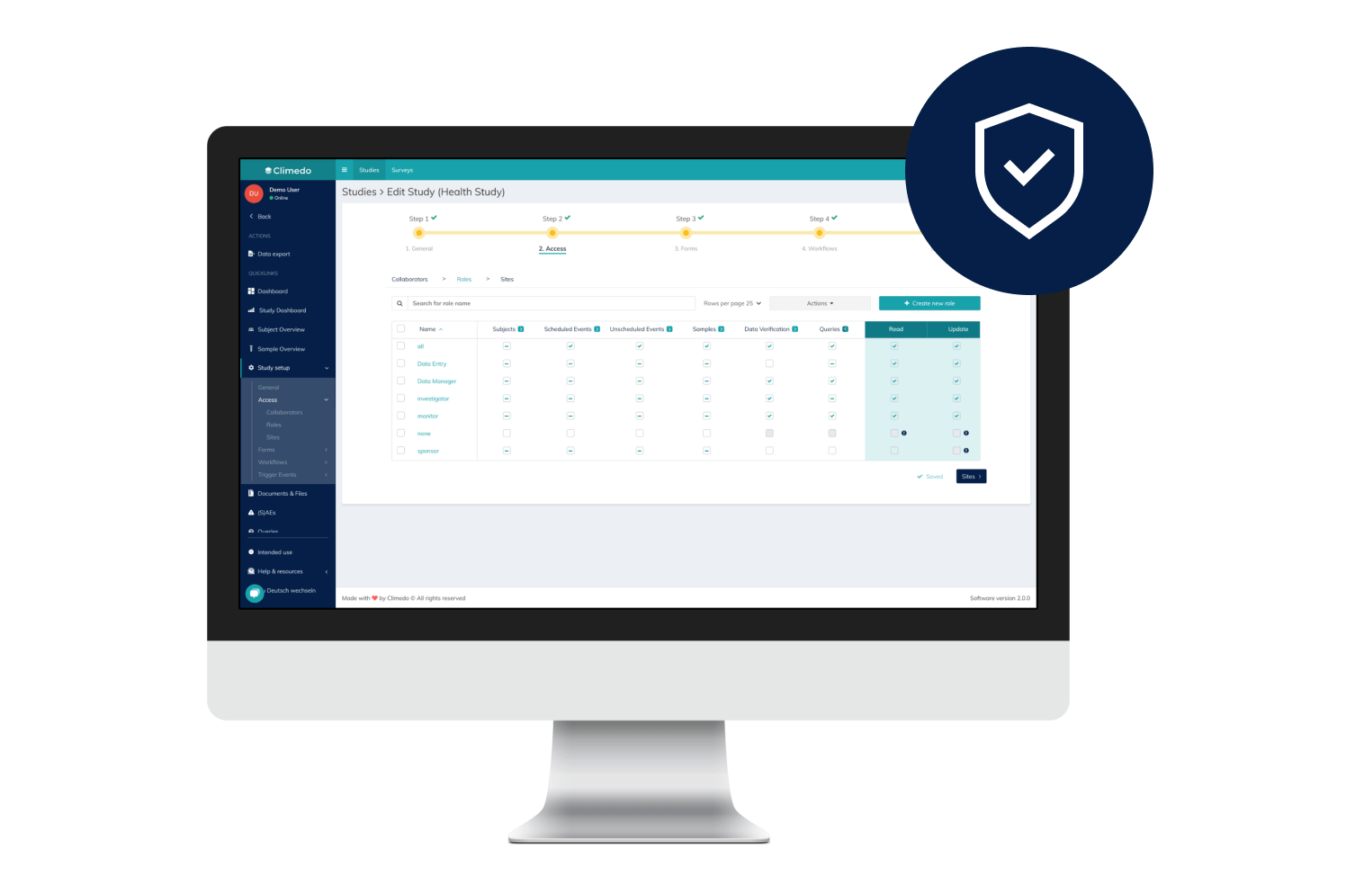
Climedo helps your hospital or research institute collect all relevant study data within a web-based, secure and user-friendly system which flexibly adapts to your processes.