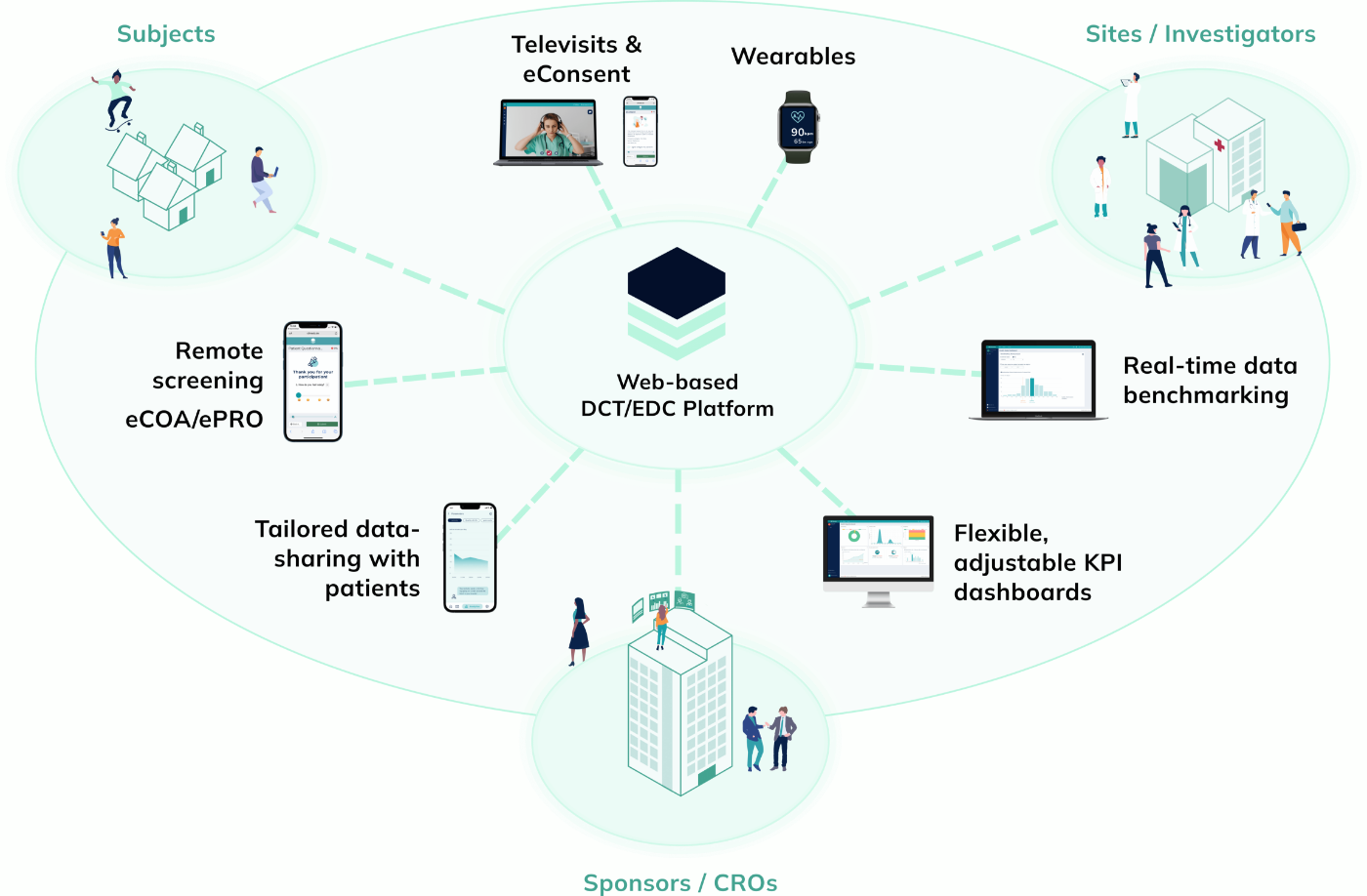
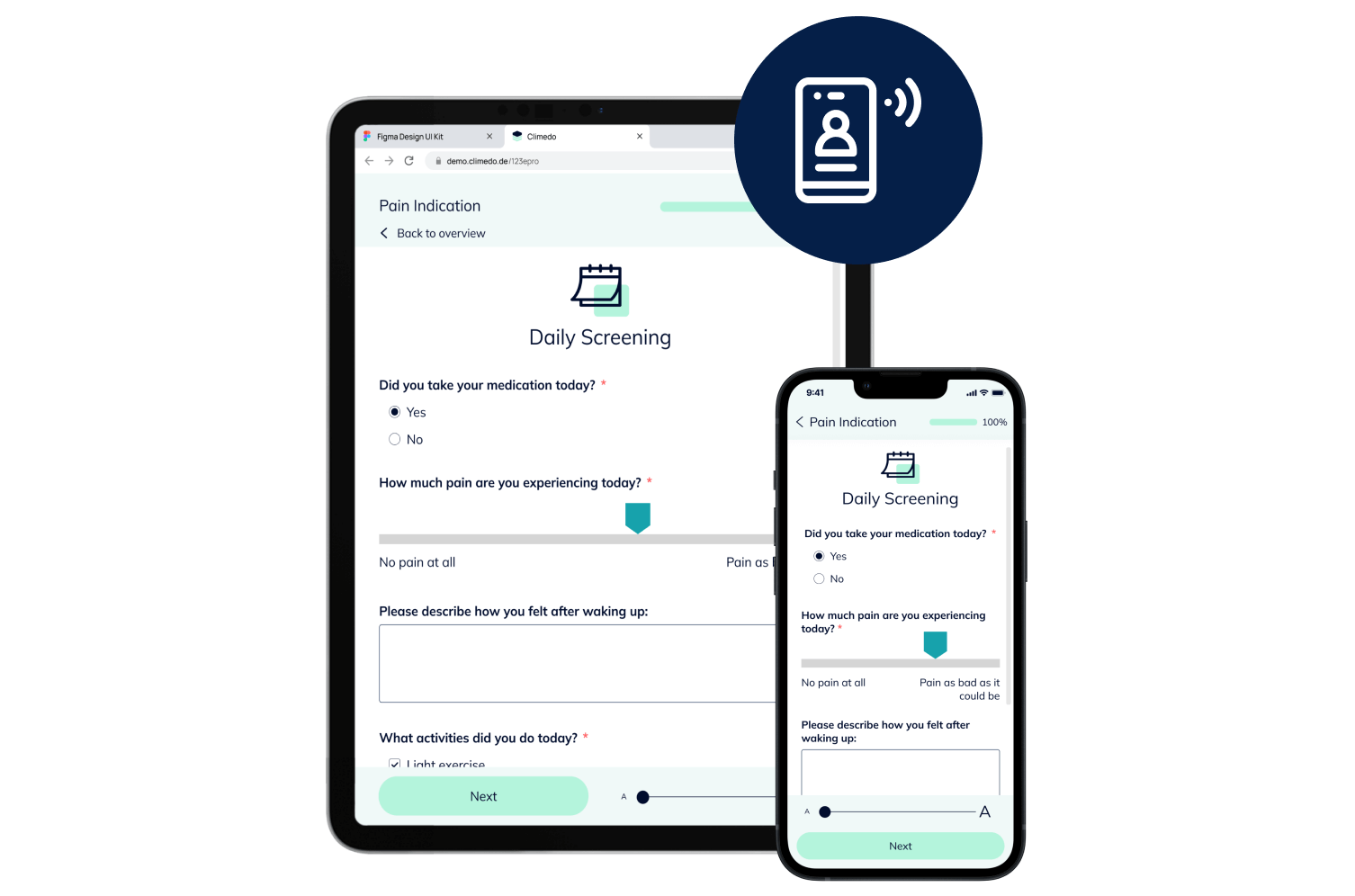
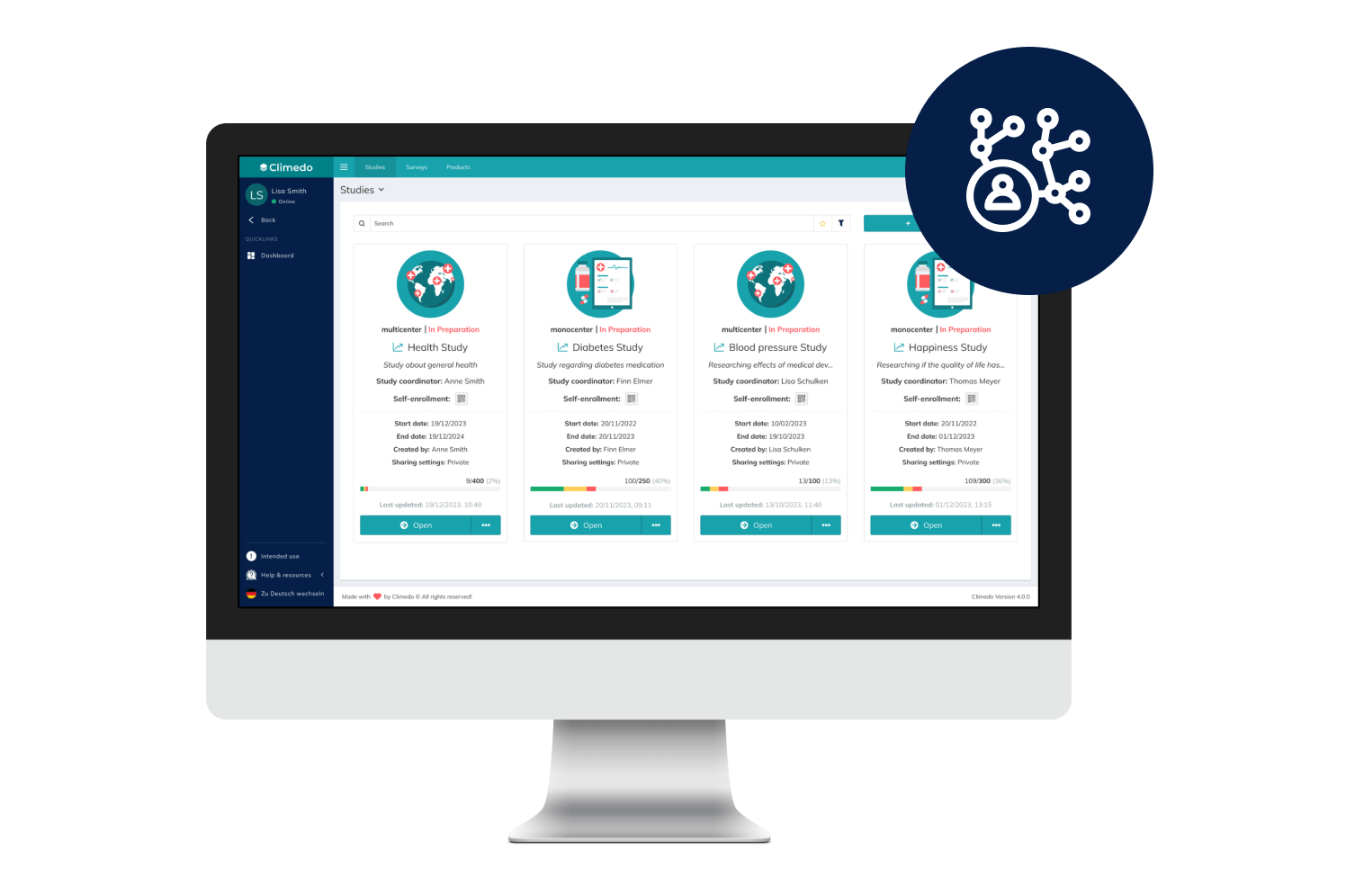
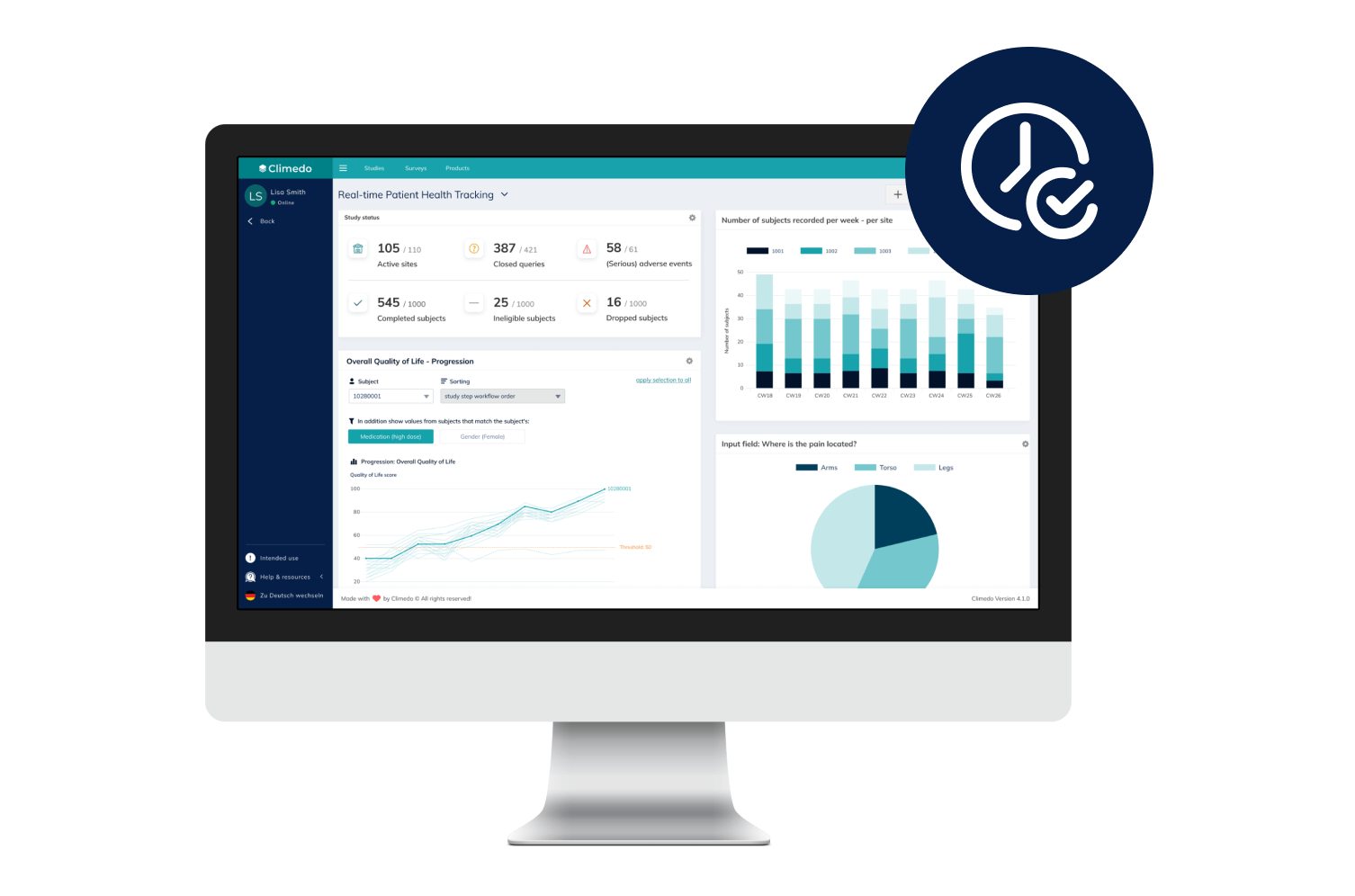
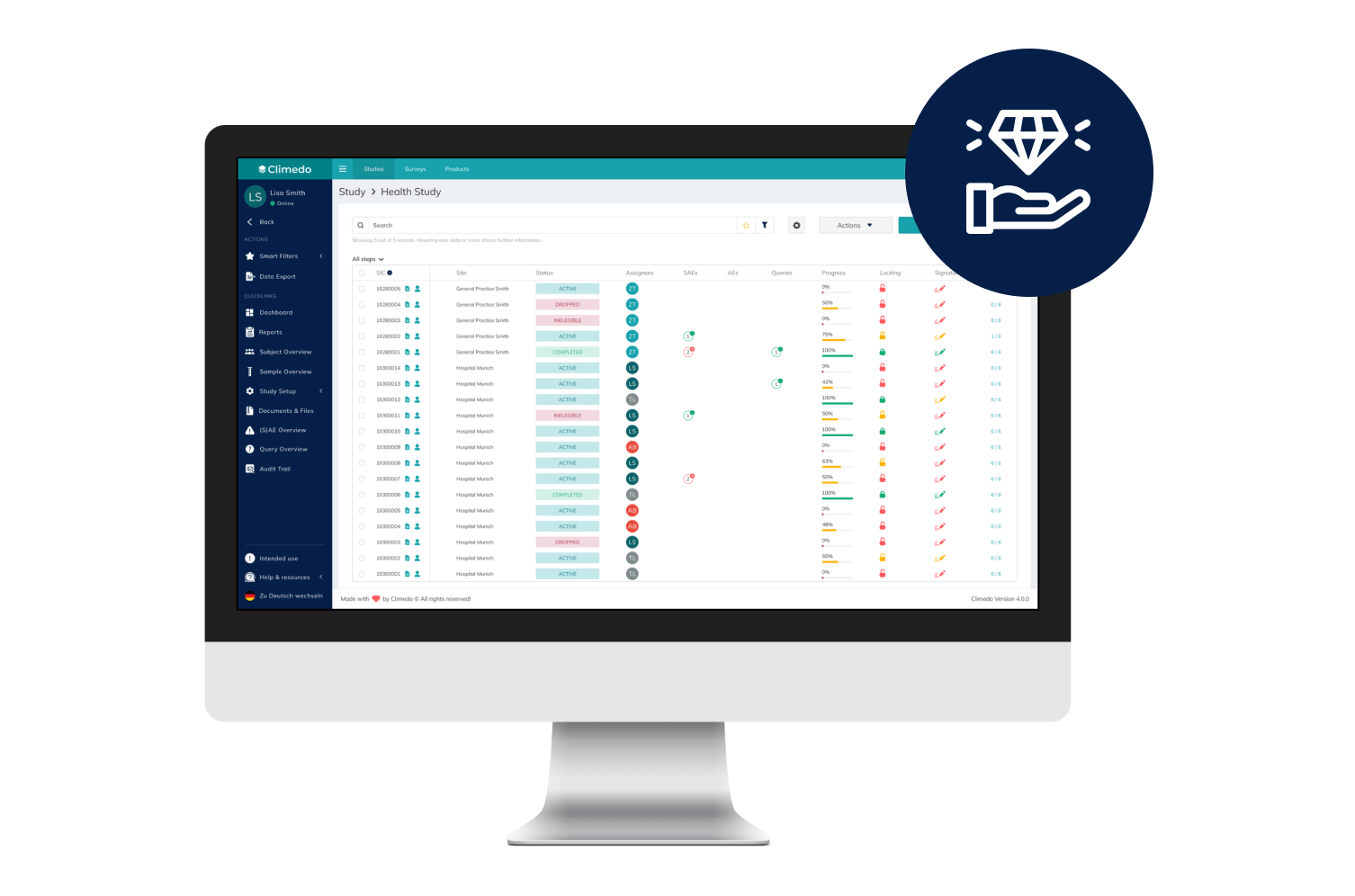
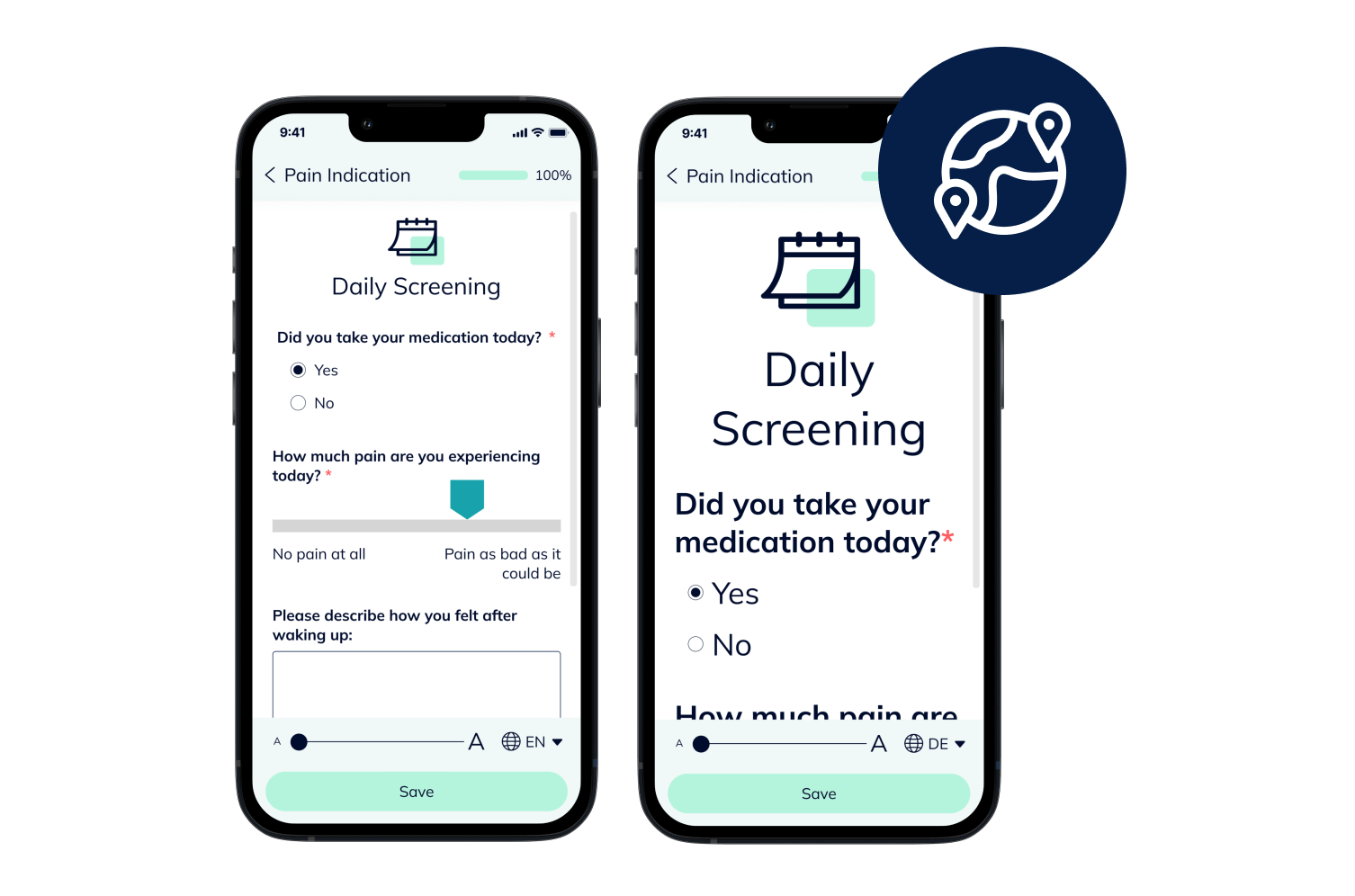
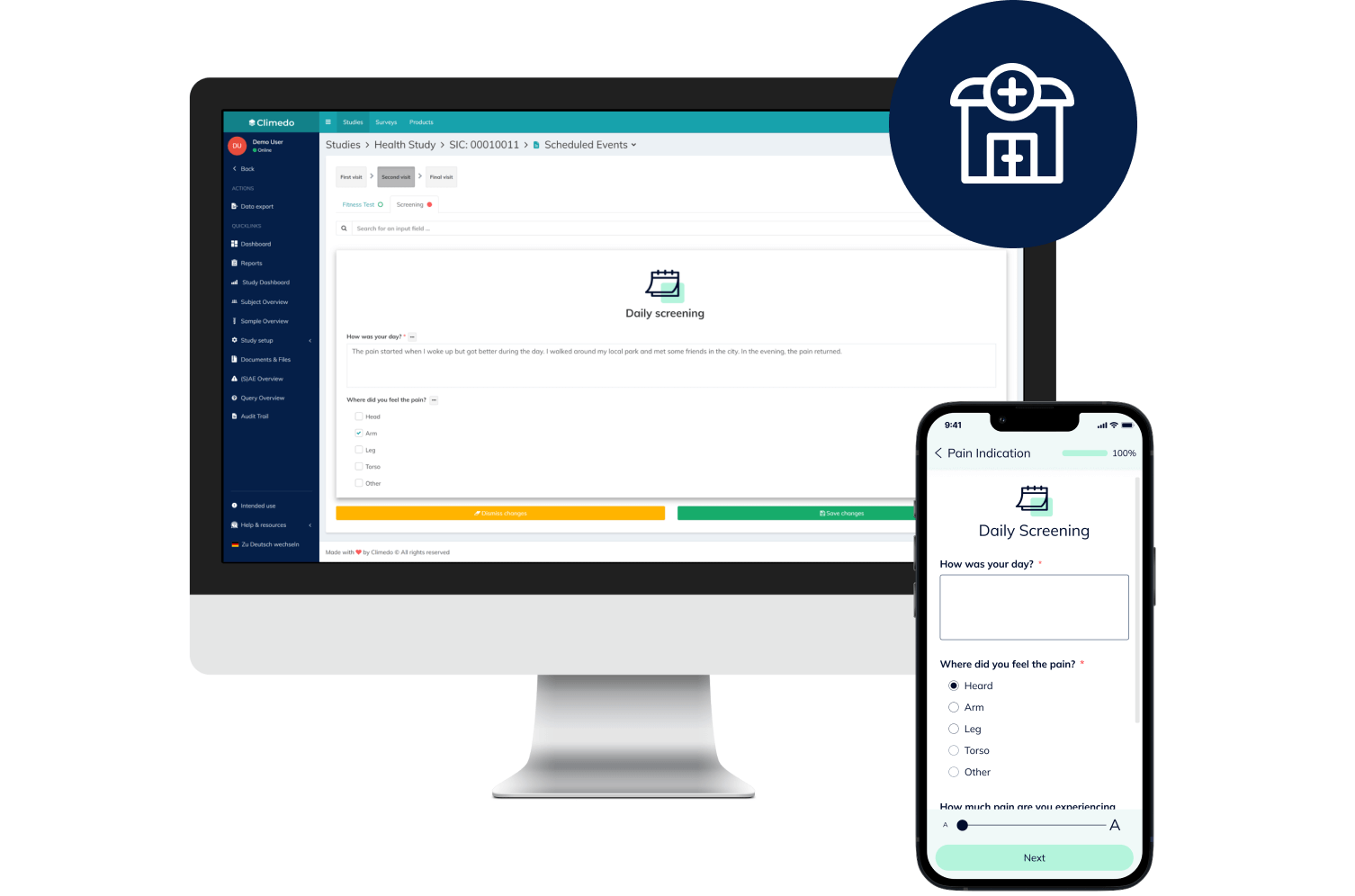
Empower Patients with Hybrid Trials
Our hybrid (or Decentralized Clinical Trials / DCT) platform enables you to implement studies in a patient-centric, flexible and decentralized manner. Enable your patients to participate remotely while saving time, costs, improving patient compliance and taking the burden off study sites.